
Mares Baby Wrap - Neopren Anzug für Babies - Girls...
15,50 €
Atlantis Onlineshop
ab 4,00 € Versandkosten

novocal Mehrzweckwagen mit beschichteten Fachböden...
705,45 €
hygi.de
Versandkostenfrei

Schöller Helios Jeans Ringel Jacke 1/2 Arm 3er Pac...
36,90 €
Sockenpara.de
Versandkostenfrei

Samshield Reithelm Miss Shield Shadowmatt Blue Shi...
649,00 €
kavalio.de
Versandkostenfrei

PURE PURE BY BAUER - Fleece-Overall WALK DOTS mit...
82,50 €
tausendkind
Versandkostenfrei

Baby Brezza Vorteils-Set: Formula Pro Mini inkl. z...
249,90 €
babybrezza.de
Versandkostenfrei

Benefit Dandelion Gold - Rouge in baby-pink mit Go...
36,00 €
Douglas DE
Versandkostenfrei

Tramontina 5-Piece Children Set Baby Friends (Spoo...
20,07 €
Knifestock.de
ab 4,50 € Versandkosten

Kinderbett ARTHUR BERNDT "Kinderbett »Lavea« 70 x...
254,99 €
baur.de
ab 5,95 € Versandkosten

CND Lecenté Create Cream Gel Polish Bloom, Baby, B...
14,60 €
HAGEL-SHOP
ab 4,99 € Versandkosten
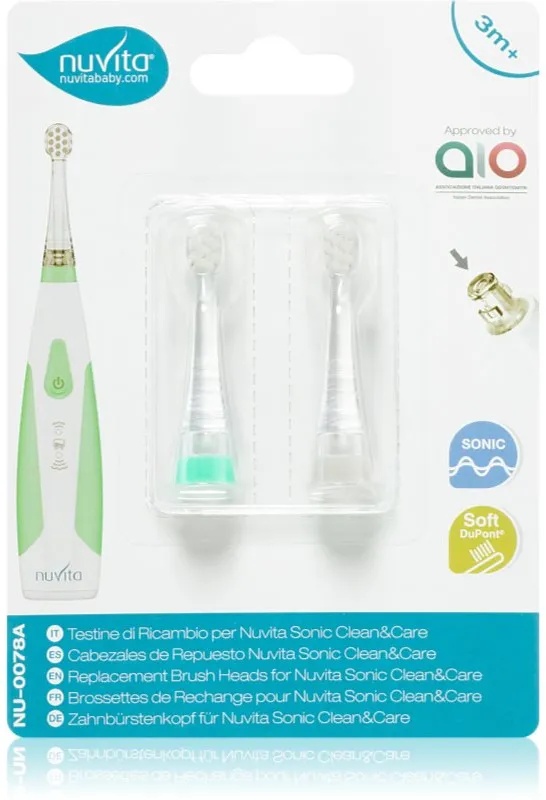
Nuvita Sonic Clean&Care Replacement Brush Heads Er...
5,01 €
NOTINO
ab 4,45 € Versandkosten

Notleuchtenakku NiMH 3,6V 4000mAh L1x3 Baby C mit...
33,75 €
AkkuShop
ab 4,50 € Versandkosten

Tchibo - 4 Baby-und-Kleinkinder-Strumpfhosen - Dun...
17,99 €
tchibo
ab 4,95 € Versandkosten

My Universe: My Baby (New Edition) (Code in a Box)...
19,35 €
Proshop.de
ab 2,99 € Versandkosten

Lorena Canals - Sea Turtle Aufbewahrungskorb, Baby...
32,00 €
connox
ab 5,94 € Versandkosten

adidas x Disney Mickey Maus Mid Hoops 3.0 Baby / K...
27,99 €
Sportspar
ab 3,95 € Versandkosten

Cloby UV Longsleeve / UV - Shirt - Größe: 6 - 12 M...
29,95 €
all-4-baby.de
ab 5,89 € Versandkosten

Tech-Protect Case for Tablet Tech-Protect Case for...
45,90 €
Galaxus.de
Versandkostenfrei

RIPNDIP Same Shit Different Day Cropped Baby T-Shi...
39,95 €
blue-tomato.com
ab 3,90 € Versandkosten

KIDIZ® Babywippe Babyschaukel mit 5 Geschwindigkei...
59,33 €
netto-online.de
ab 5,95 € Versandkosten